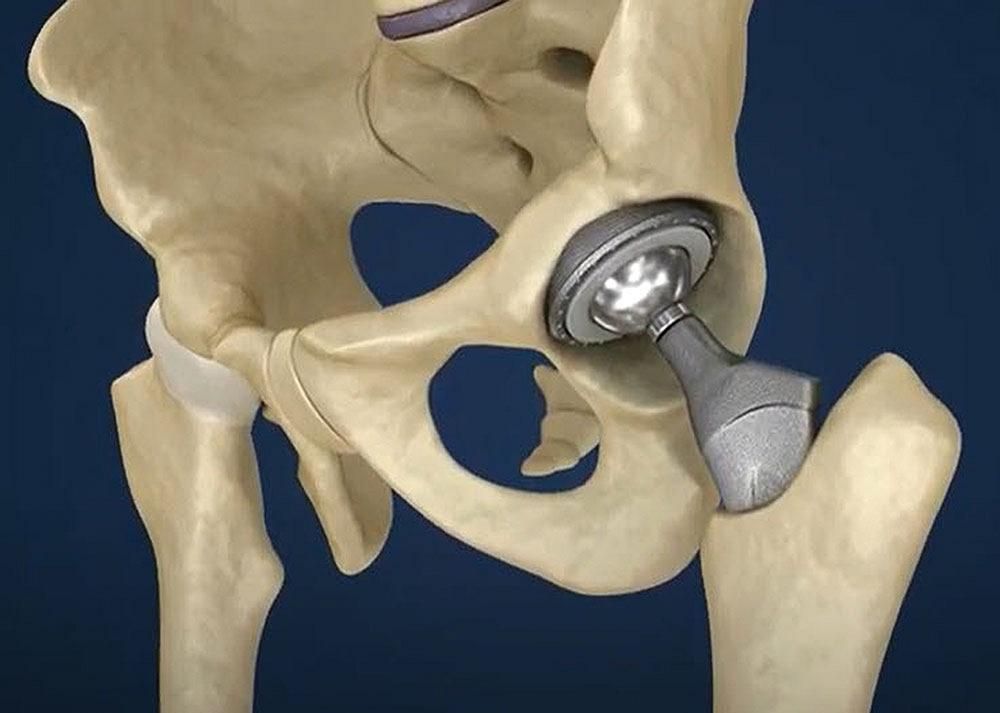
Femoral component manufacturing plays a central role in modern orthopedic care, especially in procedures such as knee replacement.
The femoral component is the part of an implant that replaces the lower end of the thigh bone (femur) in joint reconstruction. It is commonly used in total knee arthroplasty, where damaged joint surfaces are replaced to restore movement and reduce discomfort.
Orthopedic implant manufacturing has evolved over decades through advances in materials science, engineering, and medical research. Today, femoral implant manufacturers design components that match human anatomy more closely, using precise production techniques. These components are part of a larger system that includes tibial and patellar elements, forming a complete knee replacement femoral component setup.
The purpose of femoral prosthesis manufacturing is to create implants that support mobility, durability, and compatibility with the human body. This involves strict quality standards, controlled production environments, and careful testing before use in clinical settings.
Importance
Femoral component manufacturing matters because it directly affects patient outcomes in orthopedic procedures. Individuals with joint damage due to aging, injury, or medical conditions often rely on implants to regain mobility and perform daily activities.
Several key aspects explain its importance:
- Improved mobility: A well-designed femoral component for knee replacement helps restore natural joint movement, making walking and standing easier.
- Durability and longevity: Modern orthopedic femoral components are designed to last for many years, reducing the need for repeated procedures.
- Patient-specific needs: Custom femoral implant manufacturing allows adjustments based on a person’s anatomy, which can improve fit and comfort.
- Healthcare system impact: Reliable femoral implant suppliers for hospitals ensure consistent availability of implants for surgical procedures.
Orthopedic device manufacturers also play a role in advancing technology, helping to address challenges such as implant wear, alignment accuracy, and compatibility with different patient profiles.
Recent Updates
In recent years, femoral component manufacturing has seen several developments driven by technology and research. These changes aim to improve precision, efficiency, and patient outcomes.
Use of Advanced Materials
Manufacturers now use materials such as cobalt-chromium alloys, titanium alloys, and specialized polymers. These materials are selected for their strength, corrosion resistance, and compatibility with the human body.
Growth of Digital Design
Computer-aided design (CAD) and simulation tools allow femoral implant manufacturers to create more accurate models. Digital workflows help in refining the shape and surface of the orthopedic femoral component before production begins.
Expansion of 3D Printing
Additive manufacturing is increasingly used in custom femoral implant manufacturing. This method enables the creation of complex shapes that match patient anatomy, particularly in cases requiring personalized implants.
Improved Surface Technologies
Surface treatments, such as porous coatings, are applied to support better integration with bone. These enhancements aim to improve implant stability and long-term performance.
Focus on Minimally Invasive Techniques
Implant designs are being adapted to support surgical approaches that require smaller incisions. This influences how the surgical implant femoral component is shaped and manufactured.
Laws or Policies
Orthopedic implant manufacturing is regulated to ensure safety and quality. In India and many other countries, medical devices such as femoral components must meet established standards before they can be used in healthcare settings.
Regulatory Authorities
In India, the Central Drugs Standard Control Organization (CDSCO) oversees medical device regulation. Femoral component suppliers and manufacturers must comply with guidelines related to safety, testing, and documentation.
Classification of Medical Devices
Orthopedic implants are classified based on risk level. A knee replacement femoral component typically falls under higher-risk categories, requiring detailed evaluation and approval processes.
Quality Standards
Manufacturers are expected to follow international quality systems such as ISO 13485. These standards cover:
- Manufacturing processes
- Risk management
- Traceability of materials
- Post-market monitoring
Import and Distribution Rules
Femoral implant suppliers for hospitals must ensure that imported devices meet local regulatory requirements. This includes proper labeling, documentation, and compliance with safety norms.
Ethical and Safety Considerations
Policies also focus on patient safety, ensuring that orthopedic device manufacturers maintain transparency in testing and reporting. Regular audits and inspections help maintain consistent standards.
Tools and Resources
Several tools and resources support femoral component manufacturing and related processes. These tools are used by engineers, researchers, and healthcare professionals.
Design and Engineering Software
- CAD platforms help create detailed models of femoral components
- Simulation tools allow testing of stress, movement, and durability
- Digital imaging systems assist in matching implant designs with patient anatomy
Manufacturing Technologies
- CNC machining ensures precise shaping of components
- Additive manufacturing supports custom femoral implant manufacturing
- Surface treatment systems improve implant integration
Testing and Evaluation Tools
- Mechanical testing machines assess strength and wear resistance
- Biocompatibility testing ensures materials are suitable for the human body
- Imaging tools help evaluate implant positioning after surgery
Informational Resources
- Medical journals provide research on orthopedic implant manufacturing
- Government health websites share regulatory updates
- Professional associations publish guidelines for femoral prosthesis manufacturing
Example Overview of Key Elements
| Component Aspect | Description |
|---|---|
| Material Selection | Use of metals and polymers for strength and compatibility |
| Design Process | Digital modeling and simulation for accuracy |
| Manufacturing Method | CNC machining and additive manufacturing |
| Surface Treatment | Coatings to improve bone integration |
| Quality Testing | Mechanical and biological testing before approval |
FAQs
What is femoral component manufacturing?
Femoral component manufacturing refers to the process of designing and producing the part of a knee implant that replaces the end of the thigh bone. It involves material selection, precision engineering, and testing to ensure safety and performance.
How do femoral implant manufacturers ensure quality?
Femoral implant manufacturers follow strict quality standards such as ISO certifications. They conduct multiple tests, including strength and biocompatibility checks, and comply with regulatory guidelines before distribution.
What is the role of a femoral component in knee replacement?
A femoral component for knee replacement forms the upper part of the artificial joint. It interacts with other implant parts to allow smooth movement and support body weight.
Are custom femoral implant manufacturing methods widely used?
Custom femoral implant manufacturing is becoming more common, especially in complex cases. It allows implants to be tailored to individual anatomy, improving fit and alignment.
Who uses products from femoral component suppliers?
Femoral component suppliers provide implants to hospitals and surgical centers. These components are used by orthopedic surgeons during procedures such as total knee arthroplasty.
Conclusion
Femoral component manufacturing is an essential part of orthopedic implant development, supporting procedures that improve joint function and mobility. Advances in materials, design tools, and production methods continue to shape how these components are created. Regulatory frameworks ensure that safety and quality remain consistent across manufacturers and suppliers. As technology evolves, the focus remains on improving compatibility, durability, and patient outcomes in knee replacement procedures.